We've published a new paper
Following our earlier September 2023 publication in Nature Communications – Transposable elements as tissue-specific enhancers in cancers of endodermal lineage – we’re proud to announce that our newest paper has been published in Developmental Cell on 25th September, titled: Single-cell epigenome analysis identifies molecular events controlling direct conversion of human fibroblasts to pancreatic ductal-like cells.
Publication highlights and abstract
Highlights:
- Induction of pancreatic cell fate from somatic cells by
lineage-specific TFs - Epigenome and transcriptome remodeling facilitates cell fate
conversion - Direct transdifferentiation occurs through transient
progenitor cell states - FI-snMultiome-seq dissects the role of individual TFs at
single-cell resolution
Abstract:
Cell fate can be reprogrammed by ectopic expression of lineage-specific transcription factors (TFs). However, the exact cell state transitions during transdifferentiation are still poorly understood.
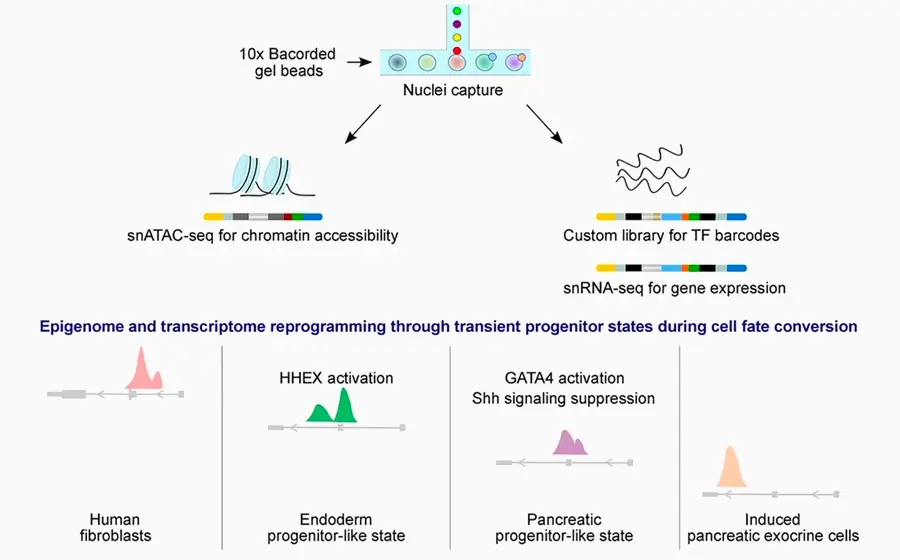
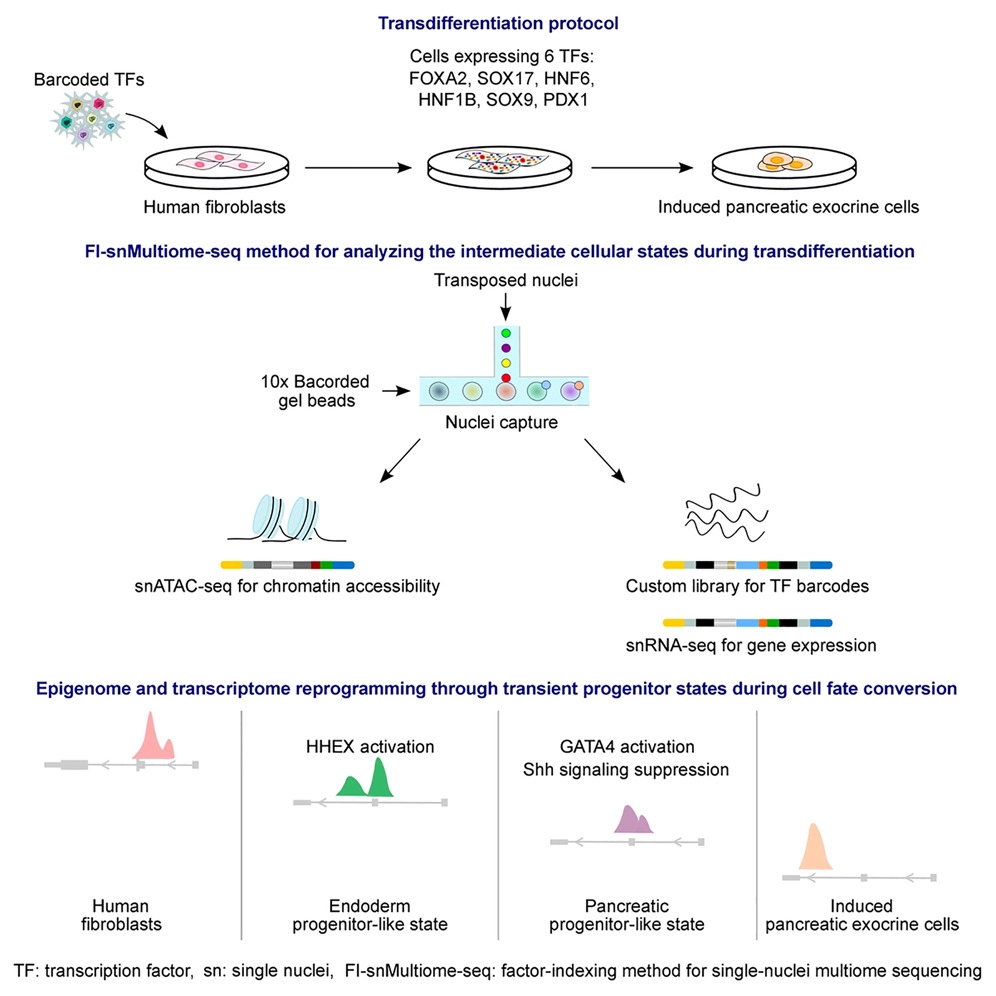
Here, we have generated pancreatic exocrine cells of ductal epithelial identity from human fibroblasts using a set of six TFs. We mapped the molecular determinants of lineage dynamics using a factor-indexing method based on single-nuclei multiome sequencing (FI-snMultiome-seq) that enables dissecting the role of each individual TF and pool of TFs in cell fate conversion.
We show that transition from mesenchymal fibroblast identity to epithelial pancreatic exocrine fate involves two deterministic steps: an endodermal progenitor state defined by activation of HHEX with FOXA2 and SOX17 and a temporal GATA4 activation essential for the maintenance of pancreatic cell fate program.
Collectively, our data suggest that transdifferentiation—although being considered a direct cell fate conversion method—occurs through transient progenitor states orchestrated by stepwise activation of distinct TFs.